Focus
Generic Pharma
Loading page...
Manufacturing
Dynapharm uses advanced manufacturing systems and strict quality controls to deliver safer, more consistent, and dependable products at scale.
Focus
Generic Pharma
Standards
Strict QC Control
Special Line
Penicillin / Cephalosporin
Overview
Workflow Snapshot
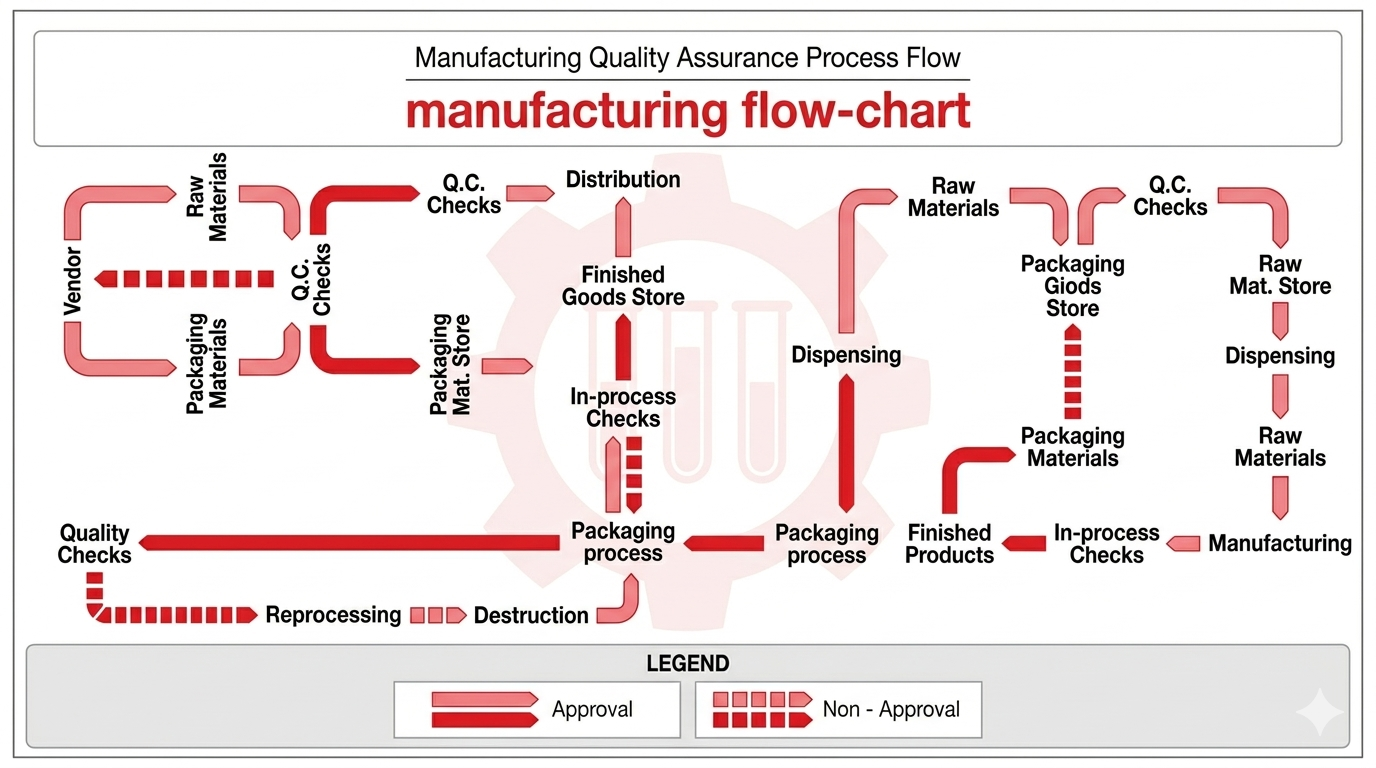
Dynapharm combines modern production systems, strict quality control, and disciplined facility planning to deliver consistent and safe pharmaceutical manufacturing outcomes.
Quality & Capability
Safety & Control
Facilities
We possess advanced facilities for the respective manufacturing processes of our products, particularly in the principal stages of introduction of functional groups, coupling and esterification, separation, and purification. Our facilities and procedures are regularly updated to accommodate industry changes and process advances.
Our manufacturing facilities support the synthesis of active ingredients and their application in drug formulations to deliver prescribed dosages for:
Process
We maintain consistent manufacturing excellence in conformity with approved industry standards across all processes involving extraction, processing, purification, and packaging of chemical materials for recognised and approved medication.
At every stage, we consistently maintain pollution prevention and contamination control measures required by the competent authorities.
Our end products meet specification-release requirements and are presented in the following forms: